A few metallic elements that are commonly found only in high oxidation states, such as niobium and tantalum, are not known to form aqua cations near the metal–nonmetal boundary, arsenic and tellurium are only known as hydrolysed species. In the highest oxidation states only oxyanions, such as the permanganate(VII) ion, MnO −Ĥ, are known. With the higher oxidation states the simple aqua ions dissociate losing hydrogen ions to yield complexes that contain both water molecules and hydroxide or oxide ions, such as the vanadium(IV) species 2+. Compounds of the metallic elements usually form simple aqua ions with the formula z+ in low oxidation states. 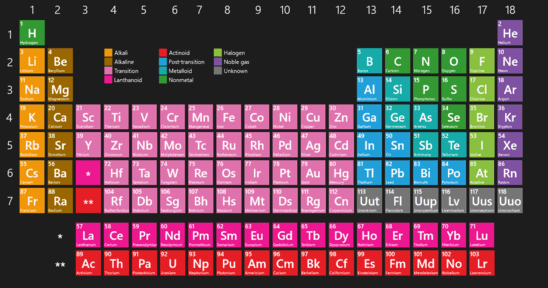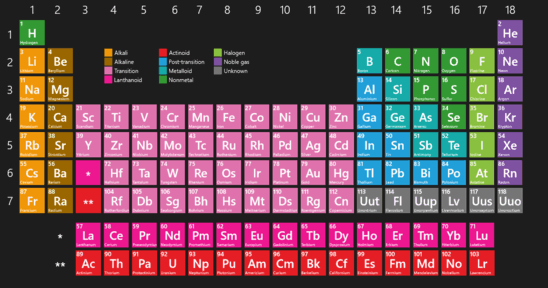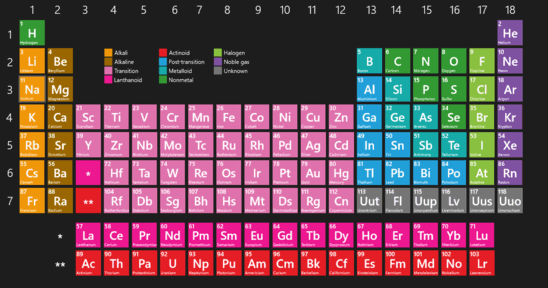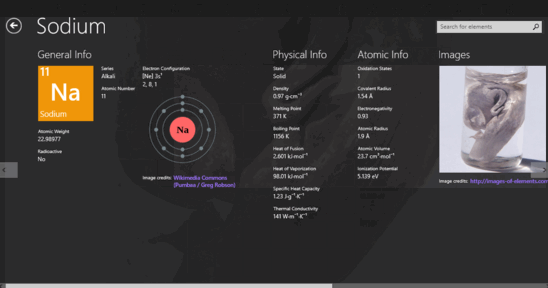 
* No experimental information regarding aqua ion structures Introduction to metal aqua ions Elements that form aqua cations Aqua ions are prominent in electrochemistry. The residence time of a molecule in the first shell varies among the chemical elements from about 100 picoseconds to more than 200 years. Water molecules in the first hydration shell exchange with molecules in the second solvation shell and molecules in the bulk liquid. The aqua ion is associated, through hydrogen bonding with other water molecules in a secondary solvation shell. The logarithm of the first hydrolysis constant is proportional to z 2/ r for most aqua ions. The strength of the bonds between the metal ion and water molecules in the primary solvation shell increases with the electrical charge, z, on the metal ion and decreases as its ionic radius, r, increases. 
Lanthanide and actinide aqua ions have higher solvation numbers (often 8 to 9), with the highest known being 11 for Ac 3+. The solvation number, n, determined by a variety of experimental methods is 4 for Li + and Be 2+ and 6 for most elements in periods 3 and 4 of the periodic table. Others are found only in compounds.Properties and behavior of hydrated cations in aqueous solutionĪ metal ion in aqueous solution or aqua ion is a cation, dissolved in water, of chemical formula z+. Some of the representative elements exist in nature as elements.Applying Concepts What does the ability of a helium-filled blimp to rise in air tell you about the density of helium? This blimp contains helium, one of the noble gases.In this periodic table, the colors of the boxes are used to classify representative elements and transition elements.Interpreting Diagrams What does the data in the square tell you about the structure of sodium atoms? This is the element square for sodium from the periodic table.How each metal is used is determined by its properties. The metals iron, copper, and aluminum have many important uses.Inferring What is the purpose for the black stair-step line? One way to classify elements in the periodic table is as metals, nonmetals, and metalloids.Interpreting Diagrams How many elements are there in the second period? In the modern periodic table, the elements are arranged in order of increasing atomic number.Observing A fourth element is grouped with chlorine (Cl), bromine (Br), and (I) iodine. In this early version of Mendeleev’s periodic table, the rows contain elements with similar properties.The numbers shown are the average atomic masses for these elements. Chlorine, bromine, and iodine have very similar chemical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
